
Fibro. Gen, Inc. Jacob, Blake Nichols, Weihua Zhang, Noelynn Oliver, David Y. Fibro. Gen, Inc. 2. Gateway Blvd., SSF, CA 9. Cardiovascular disease is the leading cause of morbidity and mortality in diabetic patients with renal disease. Both atherosclerosis and increased vascular permeability represent significant pathologies in this context. CTGF has been shown to be required to induce all forms of persistent fibrosis, and also has been implicated as a major factor involved in the pathogenesis of cardiovascular complications of diabetes. Diabetic patients undergo arterial stiffening, which has been implicated as an independent risk factor in cardiovascular mortality. To investigate the effects of FG- 3. Ab reactive to human and rodent CTGF currently in clinical trials) on diabetic vascular complications, diabetes was induced in rats by injection of STZ. Animals were treated with control Ig. G, FG- 3. 01. 9 or captopril. Diabetic rats treated with Ig. G or captopril demonstrated increases in arterial stiffness compared to non- diabetic control animals. Specifically, force- pressure curves from carotid arteries of diabetic animals demonstrated increased axial stiffness of the arteries in Ig. G and captopril treated animals. Pressure- diameter curves demonstrated increased circumferential stiffness of the carotid arteries in diabetic animals treated with Ig. G or captopril. In contrast, both axial and circumferential stiffness of the carotid arteries of diabetic animals treated with FG- 3. Morphologic evaluations of arterial sections demonstrated an increase in adventitial thickening in diabetic rats, which was prevented by treatment with FG- 3. In addition, FG- 3. Epo Side Effects Anemia Diets  
In summary, treatment with FG- 3. Since these benefits were not seen with captopril, FG- 3. ACEi and may provide a novel therapeutic approach to treating micro and macrovascular complications associated with diabetes in a clinical setting. J Clin Oncol 3. 0: 2.  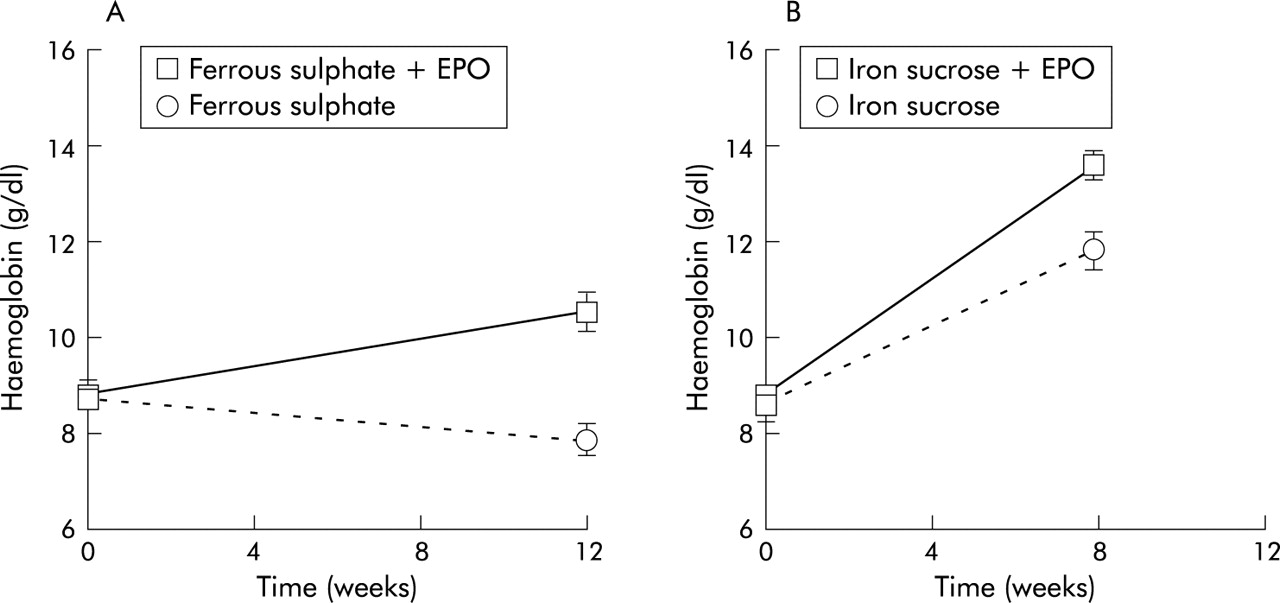
VJ Picozzi. 1, JM Pipas. AC Koong. 3, AJ Giaccia. N Bahary. 4, S Krishnamurthi. CD Lopez. 6, PJ O’Dwyer. K Modelska. 8, V.

Mircera side effects. Get emergency medical help if you have signs of an allergic reaction to Mircera: hives, sweating; fast heartbeats; difficult breathing. Anemia occurs from a lack of red blood cells. Learn about the causes, symptoms and treatments of anemia and how it relates to Chronic Kidney Disease (CKD). What is Uremia? Uremia (also called uremic syndrome) is a condition in which the urine enters the bloodstream, instead of being eliminated from the body. 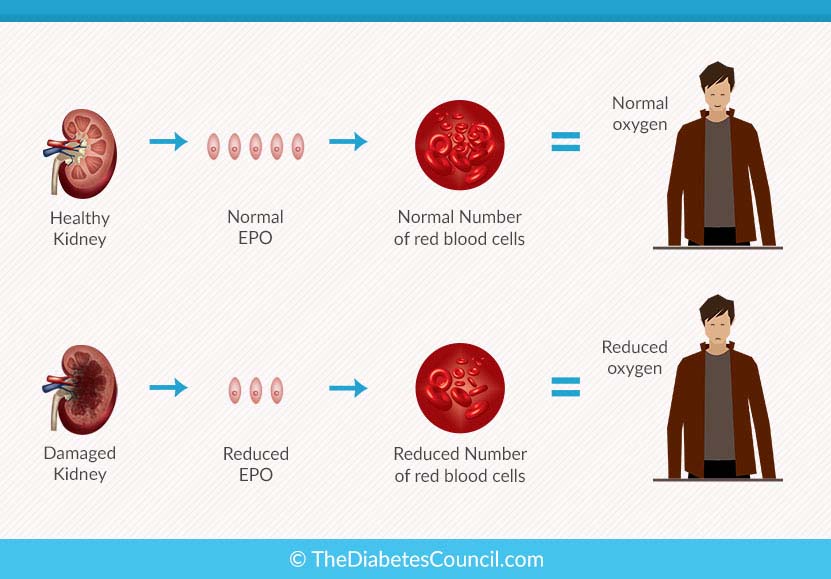  Poolman. 8, J Chou. M Zhong. 8, S Porter. T Neff. 8, F Valone. Virginia Mason Medical Center; 2 Dartmouth Medical Center; 3 University of Stanford Medical Center; 4 University of Pittsburg Medical Center; 5 University Hospitals Case Medical Center; 6. Oregon Health & Science University; 7. University of Pennsylvania Health System and 8. Fibro. Gen, Inc, San Francisco, CABackground: Connective tissue growth factor (CTGF) is overexpressed in PDAC and facilitates local desmoplasia, tumor survival and metastasis. FG- 3. 01. 9 is a CTGF- specific monoclonal antibody that decreases tumor growth and metastases and prolongs survival in orthotopic and KPC mouse models. This study evaluates safety and efficacy of FG- 3. PDAC. Methods: In an ongoing open- label, dose- escalation study, FG- 3. PDAC. Cohort 7 received 3. Day 1 and then 1. QW. Cohort 8 received 4. Day 1 and then 2. QW. Results: 7. 5 patients were enrolled at 7 centers. Baseline data: Stage III= 1. Stage IV=6. 0; ECOG=0 (n=3. ECOG=1 (N=4. 3). No SAEs or DLTs related to FG- 3. In per protocol population (n=6. PFS and OS were 4. Baseline plasma CTGF levels correlated inversely with PFS and OS (p=0. ITT population). Median FG- 3. Cmax and Cmin increased linearly with dose. Because of considerable overlap between subjects across cohorts, outcomes are correlated with drug exposure. OS, but not PFS, correlated with exposure after the first FG- 3. Day 1 Cmax, p=0. 0. Day 1. 5 Cmin, p=0. ITT population). 4. CA1. 9. 9. The magnitude of reduction correlated with Day 1 Cmax (p=0. CTGF (p=0. 0. 1). As FG- 3. 01. 9 accumulated over time, minimum FG- 3. Cmin) of 1. 50 ug/m. L on Day 4. 3 appeared to be a threshold. Median OS was 7. 7 and > 8. Day 4. 3 Cmin < 1. Day 4. 3 Cmin > 1. Conclusion: FG- 3. Interim results in this open- label study suggest OS improves with increasing exposure to FG- 3. Preclinical and Clinical Proof of Concept (POC) for Treatment of IPF with Anti- CTGF Antibody FG- 3. Porter, S., et al. Huber. 2, Kenneth E. Lipson. 1, Jonathan Goldin. Ming Zhong. 1, Thomas Neff. Frank Valone. 1, John L. Stauffer. 1 1. Fibro. Gen, Inc., San Francisco, CA, U. S. A.; 2. Deutsches Krebsforschungszentrum (dkfz), Heidelberg, Germany; 3. Department of Radiology, David Geffen School of Medicine, Los Angeles, CA, U. S. A. Introduction: The matricellular protein, connective tissue growth factor (CTGF), is a central mediator of tissue remodeling and is elevated in patients with IPF, suggesting a key role for CTGF in lung remodeling diseases. FG- 3. 01. 9, a monoclonal antibody to CTGF, reverses tissue remodeling in animal models. A radiation- induced fibrosis model strengthened preclinical POC and prompted a Phase 2 trial of FG- 3. IPF patients. Methods: Mouse lungs were injured with a single, full thorax irradiation. FG- 3. 01. 9 treatment began 1. HRCT). Gene expression and histological analyses were performed 1. In an ongoing, open- label, Phase 2 clinical trial, the initial cohort of patients with moderate to severe IPF of . Pulmonary function tests and HRCT were performed every 3 and 6 months, respectively. Results: In the preclinical model, FG- 3. Preliminary clinical data show stable or improved FVC in many patients and improved quantitative fibrosis score in 6 of the first 1. These effects appear to be related to baseline disease severity. FG- 3. 01. 9 was well tolerated, with no drug- related SAEs reported. Conclusions: Preclinical and clinical experience with FG- 3. Phase 2 IPF trial. Phase 2 Trial of FG- 3. Anti- CTGF Monoclonal Antibody, In Idiopathic Pulmonary Fibrosis (IPF): Preliminary Safety and Efficacy Results. Raghu G., et al. 5. Ganesh Raghu. 1, Mary Beth Scholand. Joao de Andrade. 3, Lisa Lancaster. Jonathan Goldin. 5, Seth Porter. Thomas Neff. 6, Frank Valone. John Stauffer. 6 1. University of Washington, Seattle, Washington, USA; 2. University of Utah, Salt Lake City, Utah, USA; 3. University of Alabama, Birmingham, Alabama, USA; 4. Vanderbilt University, Nashville, Tennessee, USA; 5. David Geffen School of Medicine, Los Angeles, California, USA; 6. Fibro. Gen, Inc., San Francisco, California, USA. Introduction: Connective tissue growth factor (CTGF) is implicated in the pathogenesis of IPF and is a potential novel therapeutic target. Objectives: To evaluate the safety, tolerability, and efficacy of FG- 3. IPF. Methods: Phase 2 prospective, open label study of FG- 3. IV every 3 weeks for 4. IPF (duration . Treatment response was assessed by changes in extent of parenchymal disease (HRCT and FVC). Results: 5. 4 subjects (males 8. FVC % predicted 6. Quantified HRCT scores of whole lung fibrosis (QLF) and all abnormal interstitial lung disease (QILD) at week 2. Changes in both QLF and QILD score were significantly correlated with changes in FVC % predicted (for QILD r=- 0. Mean decreases in FVC % predicted were less than in historical controls. Safety findings to date include 1. SAEs (none drug- related), 1 acute exacerbation, 9 respiratory- related hospitalizations, and 3 deaths (all related to IPF). Conclusions: FG- 3. IPF. No drug- related SAEs have been reported to date. Promising results of measurement of quantified lung fibrosis scores and FVC warrant pursuing the clinical trial with a higher dose of FG- 3. IPF. A Phase I Trial of the Monoclonal Antibody FG- 3. Connective Tissue Growth Factor (CTGF) in Locally Advanced or Metastatic Pancreatic Cancer. Heestand, G., et al. FG- 3. 01. 9 is a fully human monoclonal antibody against CTGF. Studies using FG- 3. Methods: This open- label, dose escalation study assessed the safety and pharmacokinetics of FG- 3. D). FG- 3. 01. 9 was initiated on D1 to assess single- agent toxicity. Standard gemcitabine and erlotinib were added on D1. Chemotherapy- naive patients with locally advanced or metastatic adenocarcinoma were eligible. Seventeen subjects (median age 6. Enrollment is ongoing in the 2. Stage 3 and 1. 3 were Stage 4. Results: No safety signals were detected with single- agent FG- 3. After beginning chemotherapy, 4 subjects experienced 7 SAEs, which were deemed unrelated to FG- 3. Nine subjects experienced Grade 3 AEs, all of which were expected in patients with pancreatic cancer. There were no Grade 4 hematological abnormalities. AEs related to gemcitabine (hematological, abnormal LFTs) and erlotinib (rash) occurred at a rate and severity consistent with the prescribing information (preliminary data). Steady state Cmax (median 4. One subject had a partial response by RECIST criteria for 9. Another subject had a minor response for 7. Three of five subjects with PET scans at baseline and D1. PET activity before starting chemotherapy. The median TTP across all cohorts was 3. CI 1. 9- 6. 2), and the median OS was 9. CI 1. 9- 1. 1. 5. Conclusions: FG- 3. Reduced PET activity after treatment with single- agent FG- 3. The CTGF antibody FG- 3. CTGF- stimulated migration of ovarian cancer cells. Samimi, G. Cancer Research July 1. Goli Samimi. 1, Carol W. Wong. 2, Suzanne M. Spong. 2 and Michael J. Birrer. 3 1. Cell and Cancer Biology Branch, National Cancer Institute, Bethesda, MD; 2. Fibro. Gen, Inc., San Francisco, CA; 3. Harvard Medical School and Massachusetts General Hospital Cancer Center Boston, MAOvarian tumors generally arise from the surface epithelium of the ovary, and require interactions with host stroma to promote tumor cell proliferation and expansion. The signaling mechanisms between tumor- associated stroma and adjacent ovarian epithelial tumor cells remain unclear. We have previously analyzed gene expression profiles of laser- capture microdissected stromal cells from normal ovary and ovarian tumors and have demonstrated that normal ovarian stroma undergoes significant gene expression changes in response to the epithelial tumor microenvironment. Connective Tissue Growth Factor (CTGF), a TGF- b- regulated gene, was identified as specifically up- regulated in tumor- associated versus normal stroma. Blood clots may happen at any time while taking EPOGEN. If you are receiving EPOGEN. These serious health problems may happen if you take EPOGEN. High blood pressure is a common side effect of EPOGEN. Your blood pressure may go up or be difficult to control with blood pressure medication while taking EPOGEN. This can happen even if you have never had high blood pressure before. Your healthcare provider should check your blood pressure often. Seizures. If you have seizures while taking EPOGEN. Your body may make antibodies to EPOGEN. Call your healthcare provider if you have unusual tiredness, lack of energy, dizziness, or fainting. You may need to stop taking EPOGEN. Serious allergic reactions can cause a rash over your whole body, shortness of breath, wheezing, dizziness and fainting due to a drop in blood pressure, swelling around your mouth or eyes, fast pulse, or sweating. If you have a serious allergic reaction, stop using EPOGEN. Tell your healthcare provider about any side effects that bother you or do not go away. You are encouraged to report negative side effects of prescription drugs to the US Food and Drug Administration (FDA). Visit www. fda. gov/medwatch or call 1- 8.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. Archives
November 2017
Categories |
 RSS Feed
RSS Feed
